PICC – CF
Evaluation of Peripherally Inserted Catheter Complications in CF Patients
According to the U.S. CF Foundation Patient Registry, more than 25% of children and 40% of adults were treated with intravenous (IV) antibiotics for flares of lung disease in 2016. Medication for these flares is often delivered through a peripherally inserted central catheter (PICC). Case series have identified important complications of PICCs in CF patients such as blood clots and infection. The frequency of PICC-associated blood clots in CF patients ranges from 2% to 8%. Catheter-related complications may interfere with completion of therapy and lead to repeated procedures and other complex medical treatments. In some cases PICC complications may discourage patients from accepting future courses of IV antibiotics. Therefore, it is very important to identify patient- and device-related factors that are linked with more frequent complications and to figure out ways to reduce these risks. Proposed risk factors fall into several broad categories. First are catheter-related factors; second are patient factors; and third are catheter-management factors.
To date, no multi-center trial has carefully studied PICC complications in a large group of adult and pediatric CF patients from the time each catheter is placed to when it is removed. The main purpose of this study is to see whether we can identify important factors in each of the three categories (patient, catheter, and catheter management) that are linked to various complications.
According to data from the U.S. CF Foundation Patient Registry (USCFFPR), more than 25% of children and 40% of adults were treated with intravenous (IV) antibiotics for pulmonary exacerbations (PEx) in 2016. This translated to 20,286 care episodes and nearly 300,000 catheter-days of risk exposure (1). Medication is typically administered through peripherally inserted central catheters (PICCs), midline catheters or totally implantable venous access devices (TIVADs) The decision to place a PICC or TIVAD in a given patient is largely a subjective one, dependent on patient and physician experiences and preferences. PICCs have become accepted as a part of usual care for pulmonary exacerbations and are generally considered safe and well-accepted by patients (2).
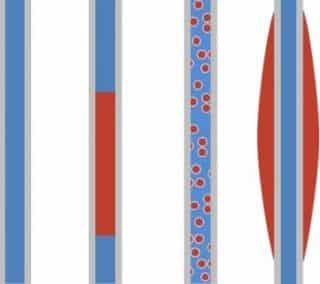
Case series have identified important complications of PICCs and TIVADs in patients such as deep venous thrombosis (DVT) and central line associated bloodstream infection (CLABSI) (3-10); central venous stenosis (11); and other types of mechanical dysfunction (12). In contrast to patients with other diseases who usually undergo PICC placement for a single treatment, however, CF patients often require repeated instrumentation over many years. This fundamental difference in how PICCs are employed confounds extrapolation of findings from studies in non-CF populations to CF patients. The incidence of PICC-associated DVT in CF patients ranges from 2 to 8% (9, 13). Catheter-related complications may delay completion of therapy and expose patients to repeated vascular access procedures and risks associated with systemic anticoagulation in cases of thrombosis. Furthermore, devastating complications, such as central venous stenosis (Figure 1), may discourage patients from subsequently accepting IV antibiotics. Therefore, it is critically important to identify patient- and device-related attributes associated with increased likelihood of complications and to devise strategies to mitigate these risks.
Proposed risk factors in CF patients fall into several broad categories. First are catheter-related factors: larger catheter size (13, 14), number of catheter lumens (13, 14), and catheter composition (10, 15). Second are patient factors: lung function (14), nutritional status (14, 16), respiratory flora (14, 17, 18), diabetes status (19-21), thrombophilia (22); and number of previously placed or guidewire-exchanged catheters (14, 23). Third are catheter-management factors: insertion technique and site selection (24-27), inpatient versus outpatient care (28), anticoagulant use (29), blood sampling practices (30), line flushing solutions (31), frequency of dressing changes (32), and duration of line use (33). The importance of many of these factors remains controversial, leading some to call for detailed prospective studies of PICC use in specific patient populations (34). To date, no multicenter trial has prospectively studied PICC and midline catheter-associated complications in adult and pediatric CF patients. We hypothesize that the rate of PICC and midline vascular complications among C Fpatients is associated with specific patient level and line level factors as well as with line insertion and management practices.
Specific Aims
Specific Aim #1: To confirm associations between patient level factors and risk of subsequent PICC and midline complications and evaluate the strength of any associations.
Specific Aim #2: To confirm associations between catheter level factors and risk of subsequent PICC and midline complications and evaluate the strength of any associations
Specific Aim #3: To confirm associations between catheter management factors and risk of subsequent PICC and midline complications and evaluate the strength of any associations
Primary Endpoint: The primary endpoint is the occurrence of vascular complications, defined as occlusion of the catheter requiring removal or symptomatic venous thrombosis in the extremity with the line as indicated by a Constans Clinical Decision Score > 2 during the time the catheter is in place (the definition does not require ultrasound confirmation, though this information will be collected, if an ultrasound is performed).
Secondary Endpoints: Bacteremia, fungemia, local phlebitis or superficial thrombophlebitis, hematoma, bleeding (including incident hemoptysis after use of thrombolytic agents), site pain, arm circumference at the level of catheter insertion, catheter fracture, temporary occlusion of the catheter cleared by thrombolytic agent, non-occlusive venous thrombosis as evidenced by ultrasound or DVT at another site, blood markers of inflammation and measures of coagulation status.
References:
- Cystic Fibrosis Foundation Patient Registry. 2016 Annual Data Report. 2017.
- Bui S, Babre F, Hauchecorne S, Christoflour N, Ceccato F, Boisserie-Lacroix V, et al. Intravenous peripherally-inserted central catheters for antibiotic therapy in children with cystic fibrosis. J Cyst Fibros. 2009;8(5):326-31.
- Cassey J, Ford WD, O’Brien L, Martin AJ. Totally implantable system for venous access in children with cystic fibrosis. Clin Pediatr (Phila). 1988;27(2):91-5.
- Ball AB, Duncan FR, Foster FJ, Davidson TI, Watkins RM, Hodson ME. Long term venous access using a totally implantable drug delivery system in patients with cystic fibrosis and bronchiectasis. Respir Med. 1989;83(5):429-31.
- Burdon J, Conway SP, Murchan P, Lansdown M, Kester RC. Five years’ experience of PAS Port intravenous access system in adult cystic fibrosis. Eur Respir J. 1998;12(1):212-6.
- Aitken ML, Tonelli MR. Complications of indwelling catheters in cystic fibrosis: a 10-year review. Chest. 2000;118(6):1598-602.
- Rodgers HC, Liddle K, Nixon SJ, Innes JA, Greening AP. Totally implantable venous access devices in cystic fibrosis: complications and patients’ opinions. Eur Respir J. 1998;12(1):217-20.
- Deerojanawong J, Sawyer SM, Fink AM, Stokes KB, Robertson CF. Totally implantable venous access devices in children with cystic fibrosis: incidence and type of complications. Thorax. 1998;53(4):285-9.
- Dupont C, Gouya H, Panzo R, Hubert D, Correas JM, Agrario L, et al. Complications of peripherally inserted central catheters in adults with cystic fibrosis or bronchiectasis. J Vasc Access. 2015;16(3):245-9.
- Chopra V, Ratz D, Kuhn L, Lopus T, Lee A, Krein S. Peripherally inserted central catheter-related deep vein thrombosis: contemporary patterns and predictors. J Thromb Haemost. 2014;12(6):847-54.
- Shin HS, Towbin AJ, Zhang B, Johnson ND, Goldstein SL. Venous thrombosis and stenosis after peripherally inserted central catheter placement in children. Pediatr Radiol. 2017;47(12):1670-5.
- Chow LM, Friedman JN, Macarthur C, Restrepo R, Temple M, Chait PG, et al. Peripherally inserted central catheter (PICC) fracture and embolization in the pediatric population. J Pediatr. 2003;142(2):141-4.
- Mermis JD, Strom JC, Greenwood JP, Low DM, He J, Stites SW, et al. Quality improvement initiative to reduce deep vein thrombosis associated with peripherally inserted central catheters in adults with cystic fibrosis. Ann Am Thorac Soc. 2014;11(9):1404-10.
- May TL, Gifford AH, Lahiri T, Black A, Trang J, Cornell AG, et al. Complications of long and intermediate term venous catheters in cystic fibrosis patients: A multicenter study. J Cyst Fibros. 2018;17(1):96-104.
- Wall C, Moore J, Thachil J. Catheter-related thrombosis: A practical approach. J Intensive Care Soc. 2016;17(2):160-7.
- Takemoto CM. Venous thromboembolism in cystic fibrosis. Pediatr Pulmonol. 2012;47(2):105-12.
- Raffini LJ, Raybagkar D, Blumenstein MS, Rubenstein RC, Manno CS. Cystic fibrosis as a risk factor for recurrent venous thrombosis at a pediatric tertiary care hospital. J Pediatr. 2006;148(5):659-64.
- Nash EF, Helm EJ, Stephenson A, Tullis E. Incidence of deep vein thrombosis associated with peripherally inserted central catheters in adults with cystic fibrosis. J Vasc Interv Radiol. 2009;20(3):347-51.
- Petrauskiene V, Falk M, Waernbaum I, Norberg M, Eriksson JW. The risk of venous thromboembolism is markedly elevated in patients with diabetes. Diabetologia. 2005;48(5):1017-21.
- Carr ME. Diabetes mellitus: a hypercoagulable state. J Diabetes Complications. 2001;15(1):44-54.
- Heit JA, Leibson CL, Ashrani AA, Petterson TM, Bailey KR, Melton LJ, 3rd. Is diabetes mellitus an independent risk factor for venous thromboembolism?: a population-based case-control study. Arterioscler Thromb Vasc Biol. 2009;29(9):1399-405.
- Balfour-Lynn IM, Malbon K, Burman JF, Davidson SJ. Thrombophilia in children with cystic fibrosis. Pediatr Pulmonol. 2005;39(4):306-10.
- Chopra V, Kaatz S, Grant P, Swaminathan L, Boldenow T, Conlon A, et al. Risk of Venous Thromboembolism Following Peripherally Inserted Central Catheter Exchange: an Analysis of 23,000 Hospitalized Patients. Am J Med. 2018.
- Fletcher SJ, Bodenham AR. Safe placement of central venous catheters: where should the tip of the catheter lie? Br J Anaesth. 2000;85(2):188-91.
- Pikwer A, Akeson J, Lindgren S. Complications associated with peripheral or central routes for central venous cannulation. Anaesthesia. 2012;67(1):65-71.
- Liem TK, Yanit KE, Moseley SE, Landry GJ, Deloughery TG, Rumwell CA, et al. Peripherally inserted central catheter usage patterns and associated symptomatic upper extremity venous thrombosis. J Vasc Surg. 2012;55(3):761-7.
- Nifong TP, McDevitt TJ. The effect of catheter to vein ratio on blood flow rates in a simulated model of peripherally inserted central venous catheters. Chest. 2011;140(1):48-53.
- Grau D, Clarivet B, Lotthe A, Bommart S, Parer S. Complications with peripherally inserted central catheters (PICCs) used in hospitalized patients and outpatients: a prospective cohort study. Antimicrob Resist Infect Control. 2017;6:18.
- Kirkpatrick A, Rathbun S, Whitsett T, Raskob G. Prevention of central venous catheter-associated thrombosis: a meta-analysis. Am J Med. 2007;120(10):901.e1-13.
- Knue M, Doellman D, Rabin K, Jacobs BR. The efficacy and safety of blood sampling through peripherally inserted central catheter devices in children. J Infus Nurs. 2005;28(1):30-5.
- Jonker MA, Osterby KR, Vermeulen LC, Kleppin SM, Kudsk KA. Does low-dose heparin maintain central venous access device patency?: a comparison of heparin versus saline during a period of heparin shortage. JPEN J Parenter Enteral Nutr. 2010;34(4):444-9.
- Gavin NC, Webster J, Chan RJ, Rickard CM. Frequency of dressing changes for central venous access devices on catheter-related infections. Cochrane Database Syst Rev. 2016;2:Cd009213.
- Chopra V, Flanders SA, Saint S, Woller SC, O’Grady NP, Safdar N, et al. The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results From a Multispecialty Panel Using the RAND/UCLA Appropriateness Method. Ann Intern Med. 2015; 163(6 Suppl):S1-40.
- Chopra V, Anand S, Krein SL, Chenoweth C, Saint S. Bloodstream infection, venous thrombosis, and peripherally inserted central catheters: reappraising the evidence. Am J Med. 2012;125(8):733-41.